Serological lipid-antibody assay for monitoring tuberculosis treatment response in children and HIV co-infected patients
Brief Description
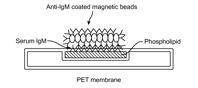
Conventional diagnosis of tuberculosis (TB) and subsequent monitoring of response to treatment requires culturing of bacteria from sputum samples, which does not always test positive. UC Berkeley investigators have developed an inexpensive serological test platform that provides accurate and rapid results for monitoring tuberculosis treatment response. This could also be used to assess end-point in drug trials. Since antibody response to pathogen is a direct reflection of bacterial burden in a host, the biomarker assay focuses on host response to the infection and treatment, as opposed to the pathogen itself. Compared to a WHO approved PCR based technology Xpert®MTB/RIF (Cepheid) used for the diagnosis of TB, which could be relatively expensive ($17,000/module and $9.98/cartridge), the present invention could provide a competitive and an inexpensive alternative for monitoring TB, especially in developing countries.
Suggested uses
- Diagnosis of TB, especially in pediatrics and HIV co-infected cases
- End point studies for TB clinical trials
- Tuberculosis biomarker assay (laboratories, pharmaceutical companies)
- Point of care (POC) diagnostic
Advantages
- Simplicity of use and higher sensitivity
- Can be used to monitor response to treatment in both adult and pediatric TB, especially in cases where inflammation is low
- Sensitive in cases of non-cavitary TB
- The reagents are widely available and inexpensive, potentially costing approximately less than $1 per test
- The level of detection is better than the standard method of examining sputum culture conversion at 2 months of treatment
- Could be used to assess the endpoint of a drug trial
Patent Status
| Country | Type | Number | Dated | Case |
| United States Of America | Issued Patent | 9,835,624 | 12/05/2017 | 2011-107 |
Contact
- Javed Afzal
- jafzal@berkeley.edu
- tel: View Phone Number.
Other Information
Keywords
Tuberculosis, HIV, Infectious disease, Antibody assay
